2019 Case Study DRX9000®
2019 Case Study DRX9000®
CASE PRESENTATION It’s all in the pictures
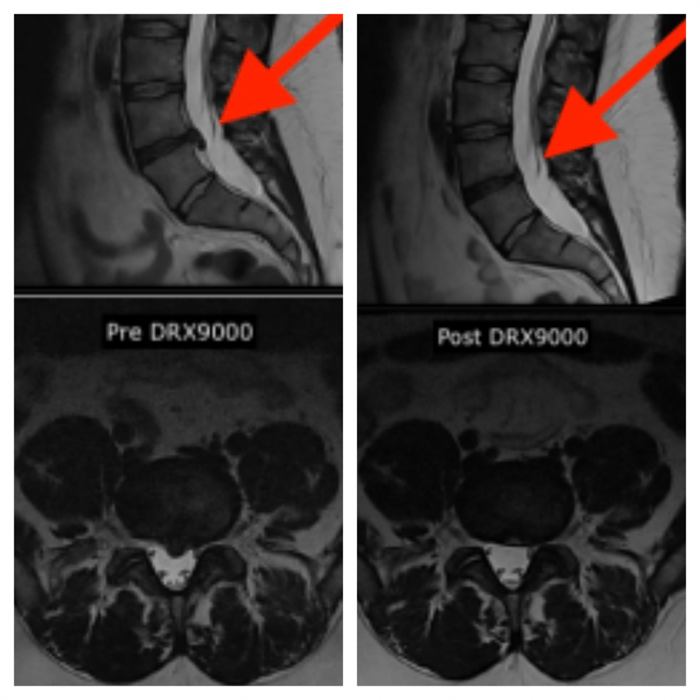
A 28-YEAR-OLD BANK TELLER presented with a one-year history of lower-back pain radiating down his left lower extremity, associated with numbness and tingling sensation down the left leg in the L4 and L5 dermatomal distribution. The symptoms were interfering with the patient’s work and sleep. Prior to his initial visit with Dr. Mustafa Hammad, he underwent six weeks of physical therapy and was treated with NSAIDs, opioids and Lyrica without any relief. A pre-treatment MRI (figure 1) revealed moderate disc herniation at L4-5 level touching the L4 and L5 nerve roots on the left The patient underwent 20 treatment sessions on the DRX9000® over a six week period. A follow up MRI, one month after the last treatment (figure 2) reflected almost total resolution of the herniated L4-5 disc, with improvement in disc morphology, disc height, and neural canal AP diameter at the L4/5 level. Clinically the patient had total resolution of his symptoms. After a one-year follow-up with the patient, he was still symptom free and a repeat MRI still showed no changes from the previous post-treatment MRI. Mustafa A. Hammad, MD, DO, DABIPP, FIPP, FACP, CPI, FAAN, owner of The NeuroMedical Institute, is boardcertified in neurology, pain medicine and interventional pain management using the DRX900. He can be reached via inewromed.com.
The Case for DRX9000®

The DRX9000® provides a program of treatment for relief from pain form those patients suffering with chronic and low-back pain. Under physician direction, each treatment consists of a prescribed treatment period on the DRX9000® to provide intermittent cycling of different distraction forces that helps relieve pressure on structures that may be causing low-back pain. Pain associated with degenerative disc disease, bulging discs, herniated discs, sciatica and posterior facet syndrome all respond well. It achieves these affects through decompression of intervertebral discs.
What results can patients expect from the DRX9000®
After only six weeks of treatment, clinical studies have shown impressive results in areas of pain relief caused by herniated, bulging, ruptured or degenerative discs, as well as posterior facet syndrome, sciatica and many cases of failed back surgeries.
Pre- and post-MRIs have shown reduction in the size and extent of herniations after six weeks of treatment with the DRX9000® . Excite Medical is a medical device company based in Tampa, FL, USA. Excite’s flagship product, the DRX9000® , is found in more than 1,000 hospitals, clinics, and private health centers located in more than 45 countries. Excite Medical, registered with the U.S. FDA, holds four FDA 510(k)s including one for the DRX9000® .
The DRX9000® has been cleared by the FDA to treat patients suffering with incapacitating lower-back pain and sciatica caused by herniated discs, degenerative discs, and posterior facet syndrome. Published research has shown the vast potential of the DRX9000® — the research was conducted by highly-respected physicians and researchers associated with the Mayo Clinic, Stanford, Duke, Johns Hopkins University, the University of California San Francisco, and other prestigious medical institutions.
DRX9000® systems are now available for as little as $700 per month lease for qualified physicians. For more information about the DRX9000® call/text +1-813-210-1000 or visit ExciteMedical.com
SOURCES:
https://irp-cdn.multiscreensite.com/9614ed0a/ files/uploaded/MRI%20Findings.pdf
https://www.ncbi.nlm.nih.gov/pmc/articles/ PMC4603263/
https://pdfs.semanticscholar.org/7524/c86438097f2777fb86632c3c45e0b3ecf796.pdf
https://www.spine-health.com/blog/ spinal-fusion-surgery-worth-cost-stenosis-patients
https://my.clevelandclinic.org/health/diseases/9516-back-pain-basics

