DRX9000® Lumbar True Spinal Decompression™ Machine
DRX9000® Machine Features
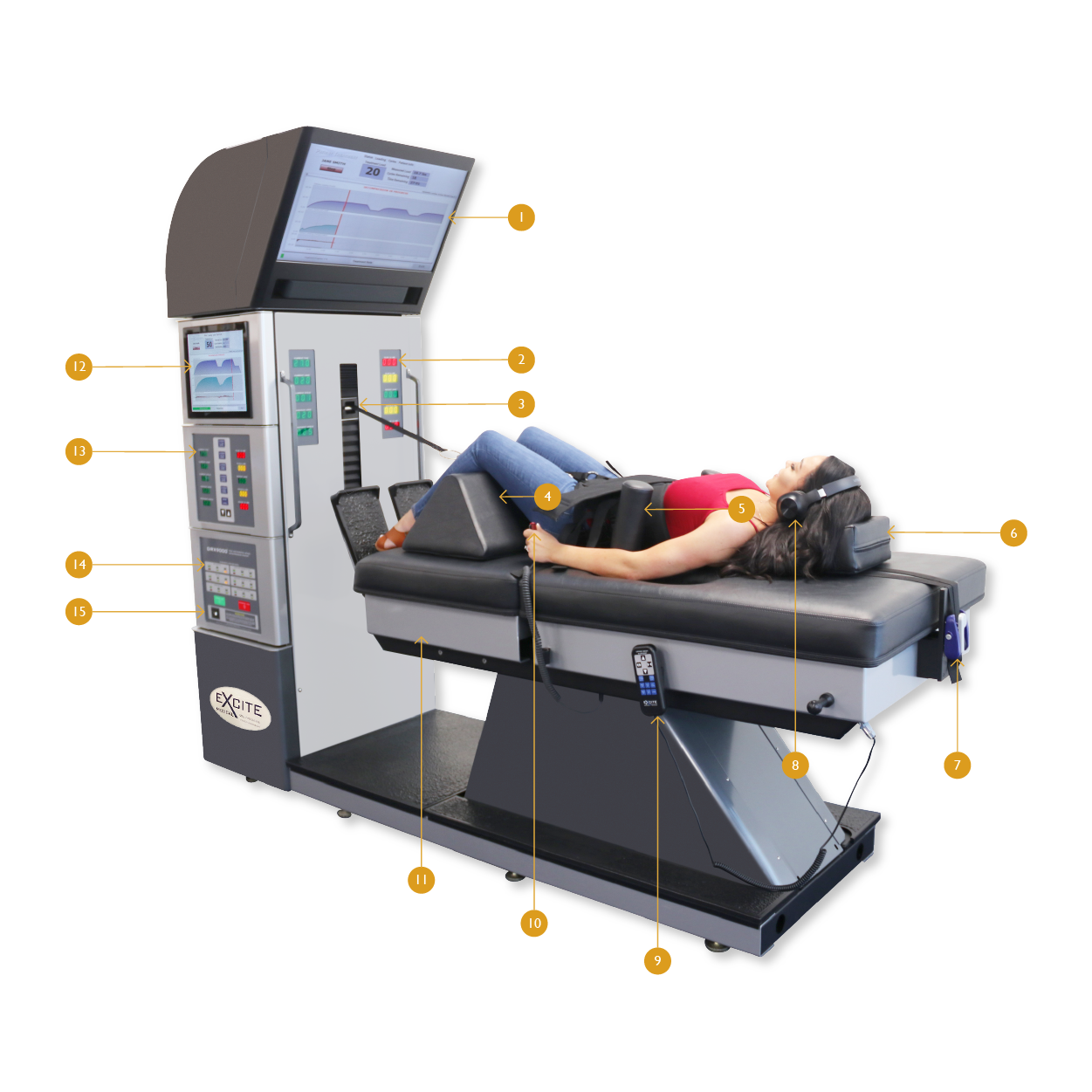
To understand DRX9000® spinal decompression machine features, please follow the numbers from the DRX9000® decompression machine image above.
The DRX9000® machine has a screen controlled by multi-media player to provide patients with relaxing music or educational videos about treatment for back pain, treatment for neck pain, herniated disc, bulging disc, sciatica or any video you choose to play..
DRX9000® treatment progress can be viewed by the patient through these displays. This will allow the patient to keep track of DRX9000® treatment progress.
DRX9000® treatment dynamics allows you to target a specific lumbar region. Back pain in patients can be caused by herniated discs, bulging discs or degenerative disc diseases in different parts of the spine. With this feature you will be able to target the specific disc level region that is causing the pain or other conditions.
DRX9000® Knee Pillow is custom designed to complement the DRX9000® treatment.
DRX9000® upper body support system
DRX9000® Head Pillow, to support head position during spinal decompression treatment
DRX9000® Harness Tensioner helps sucure the upper body harness as True Spinal Decompression™ takes place.
DRX9000® operator remote
DRX9000® patient safety switch
DRX9000® floating lower mattress
DRX9000® touch screen computer to operate the device
DRX9000® Lumber leveler selector
DRX9000® system diagnostics
DRX9000® System power ON/OFF button
The DRX9000® decompression machine Dimensions are:
Length ……………. (Overall) 113.00 in (288cm)
Width ………………. 32.50 inches (83cm)
Height ……………… 85.50 inches (218cm)
Weight …………….. 1,280 lbs. (580 kg)
Crated Weight…… 1,730 lbs. (784 kg)The Recommended Room Requirement for a is 8’x10’ (2.5m x 3.1m)
Conditions that can be treated with DRX9000® decompression machine treatment
The DRX9000® True Non-Surgical Spinal Decompression® treatment is specifically engineered to provide relief of pain, sciatica and other symptoms associated with Herniated Discs, Bulging Discs, Degenerative Disc Disease and protruding discs.
DRX9000® decompression treatment
The DRX9000® True Non-surgical spinal decompression® treatment for back pain has been designed to aid healthcare providers in their efforts in treating low back (lumbar) pain. DRX9000® and DRX9000C® treatments provide relief to patients suffering from chronic back pain or neck pain. The DRX9000® spinal decompression machine has been cleared by the FDA to treat herniated discs, bulging discs, degenerative disc disease, and posterior facet syndrome. The DRX9000Ò is the only system available in its field to utilize a servo motor; in doing so, the DRX9000® contains nested closed-loop feedback that ensures that the therapy stays true to each patient’s logarithmic spinal decompression treatment.
How DRX9000® decompression machine treatment works
DRX9000® system works on the injuries that happen due to nerve compression by applying equal amount of pressure on the vertebrate columns, by stretching the spine, this creates a gap in between the discs, and with improved flow of nutrients through both the vertebrate columns of spine the injuries heal slowly and steadily after regular DRX9000® decompression treatment sessions, as suggested by your doctor. Many patients are living better and happier lives.
DRX9000® Spinal Decompression Machine Treatment FAQ
How long does it take to see results after non surgical spinal decompression DRX9000® treatment?
Is non surgical spinal decompression DRX9000® treatment successful?
What are the contraindications for the DRX9000® or criteria that determine who is not a candidate for DRX9000® true non-surgical spinal decompression therapy?
- Pregnancy
- Prior Lumbar Fusion
- Metastatic Cancer
- Sever osteoporosis
- Spondylolistheses (unstable)
- Compression fracture of the lumbar spine below L-1 (recent).
- Pars Defect
- Pathologic Aortic aneurysm
- Pelvic or abdominal cancer
- Disc space infections
- Sever peripheral neuropathy
- Hemiplegia, paraplegia, or cognitive dysfunction
How much Time does it take to complete DRX9000® treatment?
Am I the right person to opt for a non surgical spinal decompression DRX9000® treatment?
What do Doctors say about DRX9000® decompression treatment?
Excite Medical is the owner of all DRX9000® associated FDA 510(k)’s. For sales, service, or support related to the DRX9000®, contact us right NOW! Or Call/Text +1-813-210-1000
DRX9000 FDA Clearance 2003 510k K022602.
DRX9000 FDA Clearance 2006 510k True Non-Surgical Spinal Decompression K060735.
